Plating is used for applying coatings for decoration or
corrosion resistance. It general plating is suitable for thin coatings
and most often not cost effective for thicker coatings.
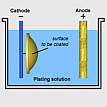
The electroplating processes are carried out in an electrolytic cell.
The cell contains a socalled electrolyte (an electrical conducting liquid)
and two electrodes. The negative electrode (cathode) is the part to be
coated and the positive (anode) is the coating metal. Under low-voltage
current, ions in the electrolyte move towards the cathode and deposit
on it.
Electroless plating enables metal coating of non-conductive materials,
such as plastics, glasses and ceramics, but is also used for metals. Unlike
electroplating, coatings usually have a very uniform distribution of thickness.
The deposition is carried out in liquids (solutions), and is based on
chemical reactions (mainly reductions), without an external source of
electric current.
Vapour deposition is a class of processes used for coating metals, plastics,
paper and glass with all kind of materials (plastics, metals, ceramics)
for decorative purposes, wear and corrosion resistance. The coating layer
is formed by condensation of metal vapour arising from molten metal, from
high voltage discharge between electrodes (sputtering), or from chemical
processes, like hydrogen reduction.
Danish Name
Category
Anodizing
processesElectro-
plating processesChromium & Hard-chromium plating
Copper & Tin plating
Plating with Gold, silver and other precious metals
Zinc & Nickel plating
Electroless
plating
processesChromating
Electroless nickel-plating
Phosphating
Interference
colouring Vapour
depositionPACVD - Plasma Assisted CVD
PVD - Physical Vapour Deposition
Vapour metalizing
Electro-formingReferencesGalvanisk Kompagni A/S
Galvaonline
BIV
Additional
informationCoating thickness in electroplating depends on the current intensity.
Irregular shapes and blind holes causes non uniform coatings. Hydrogen
embrittlement of parts can be a problem.
Electroplating works similar to electroplating but much thicker layers
are made so the surface layer becomes a part itself.
Electroless plating can be done using two different process principles.
Autocatalythic-plating is used for nickel, copper and their alloys. For
tin plating of copper and aluminium the ion exchange-plating (very thin
coatings) is used.
All vapour processes involve treatment in a vacuum chamber. The equipment
is big, heavy and expensive and requires maintenance. Limitations in the
size of the objects to be coated is imposed by the capital expenditure.
Environmen-
tal notes Photo
Copyright
This page is part of Design inSite